Nobivac® L4 Questions and Answers
-
What are the indications for Nobivac L4?
For active immunisation of dogs against L. interrogans and L. kirschneri:
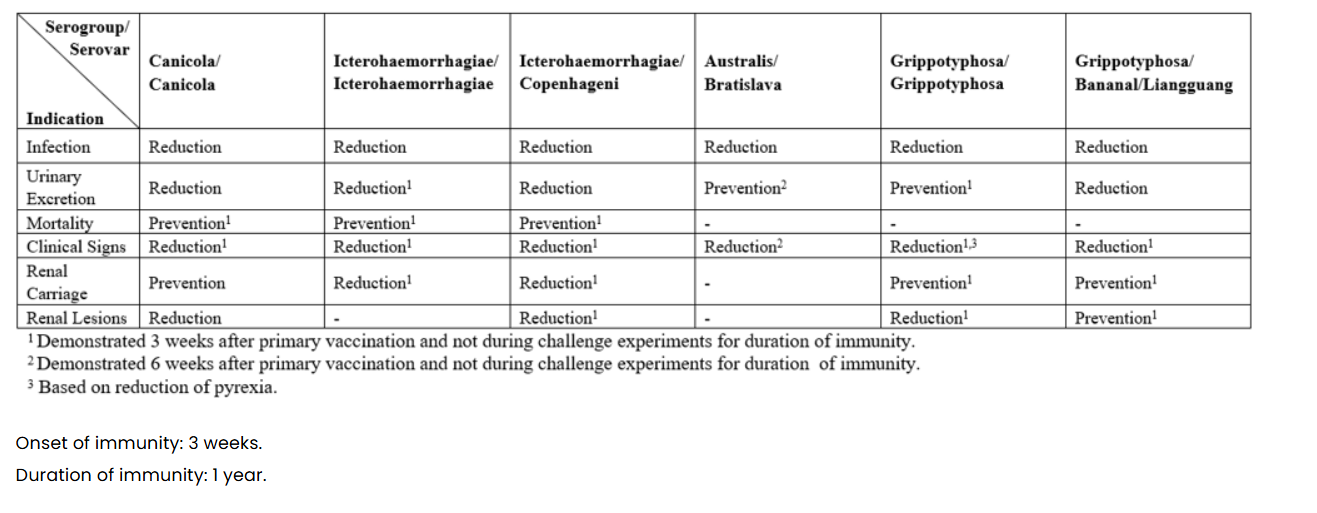
-
What is the interval for Nobivac L4?
Administer two vaccinations of 1 dose (1 ml) of vaccine with an interval of 4 weeks to dogs from 6 weeks of age onwards.
The first vaccination can be administered from 6 to 9* weeks of age and the second vaccination from 10 to 13 weeks of age.
*In cases of a high level of MDA, first vaccination is recommended at 9 weeks of age.
-
What is the duration of immunity for Nobivac L4?
Duration of immunity: 1 year.
Revaccination: Dogs should be re-vaccinated annually with one dose (1 ml) of vaccine.
-
What is the onset of immunity for Nobivac L4?
The onset of immunity following the second vaccination of the primary course is 3 weeks.
-
Can Nobivac L4 be given at the same time with other Nobivac vaccines?
Safety and efficacy data are available which demonstrate that this vaccine can be mixed and administered with vaccines in the Nobivac range containing canine distemper virus, canine adenovirus type 2, canine parvovirus (strain 154) and/or canine parainfluenza virus components for subcutaneous administration. The product information of the relevant Nobivac vaccines should be consulted before administration of the mixed product. When mixed with these Nobivac vaccines, the demonstrated safety and efficacy claims for Nobivac L4 are no different from those described for Nobivac L4 alone. When mixed with Nobivac vaccines containing canine parainfluenza virus at annual revaccination, it has been established that there is no interference with the anamnestic response induced by the injectable canine parainfluenza virus component.
Safety and efficacy data are available which demonstrate that this vaccine can be administered on the same day but not mixed with vaccines in the Nobivac range containing Bordetella bronchiseptica and/or parainfluenza virus components for intranasal administration. Safety data are available which demonstrate that this vaccine can be administered at the same time but not mixed with the inactivated vaccine in the Nobivac range against Bordetella bronchiseptica. When this vaccine is administered in association with the inactivated vaccine in the Nobivac range against Bordetella bronchiseptica the demonstrated antibody response data and other immunity data of this vaccine are the same as when this vaccine is administered alone.
Safety and efficacy data are available which demonstrate that this vaccine can be administered at the same time but at different administration sites with vaccine in the Nobivac range against rabies, although this may, in some instances, lead to vomiting.
For simultaneous use 1 dose of a Nobivac vaccine containing canine distemper virus, canine adenovirus type 2, canine parvovirus, and/or canine parainfluenza virus components should be reconstituted with 1 dose (1ml) of Nobivac L4. The mixed vaccines should be at room temperature (15oC - 25oC) before they are administered by subcutaneous injection. If Nobivac Rabies is given concurrently with Nobivac L4 then this should not be mixed but should be given as a separate subcutaneous injection at a separate site.
-
Can Nobivac L4 be used in pregnancy?
Nobivac L4 can be used during pregnancy.
We do not hold specific data regarding use during lactation and therefore cannot recommend this use during this period.
-
Are there any breed contraindications for Nobivac L4?
There are no breed specific contraindications for Nobivac L4.
-
What are the storage requirements for Nobivac L4?
Nobivac L4 requires storage in a refrigerator between 2 oC and 8 oC. Do not freeze. Protect from light.
-
Are Nobivac vaccines inactivated or live vaccines?
Live attenuated vaccines
Live vector vaccines
Killed (inactivated) vaccines
Inactivated sub-unit vaccines
Nobivac DHP
Nobivac Myxo-RHD PLUS Nobivac L4
Nobivac FeLV
Nobivac DHPPi
Nobivac Rabies
Nobivac Respira Bb
Nobivac Ducat
Nobivac KC
Nobivac Parvo-C
Nobivac Pi
Nobivac Tricat Trio
-
What is the minimal dose for vaccinations?
Each vaccine is formulated on the basis of a minimum immunising dose, not a dose per body weight. Therefore the full vaccine dose/reconstituted amount is the minimal immunising dose.
-
What are the storage requirements for Nobivac vaccines?
In order for the product shelf-life to be as it is stated on the product's SPC it should be stored as per the datasheet's storage requirements. This will always be our recommendation for product use.
In the case of any doubt it would be advisable to contact the Technical Product Support team before discarding any product.
In the event where it is advisable to discard product it may be possible to claim on the practice's insurance. Our Technical Product Support team can write a letter of support for insurance claims, pertaining to deviations in storage conditions, should you require it.
Product images are representations of packaging and may not accurately reflect the current packaging available in the GB marketplace.
Date of preparation: February 2026
Not answered your question?
Email your question and our team will reply within 2 business days.
Contact our Technical Support Team today.

Nobivac® DHP contains live canine distemper virus (strain Onderstepoort), canine adenovirus 2 (strain Manhattan LPV3) and canine parvovirus (strain 154). POM-V.
Nobivac® DHPPi contains live canine distemper virus (strain Onderstepoort), canine adenovirus type 2 (strain Manhattan LPV3), canine parvovirus (strain 154) and canine parainfluenza virus (strain Cornell). POM-V.
Nobivac® Ducat contains live attenuated feline rhinotracheitis virus (strain G2620A) and feline calicivirus (strain F9). POM-V.
Nobivac® FeLV is an inactivated subunit vaccine containing purified p45 FeLV-envelope antigen. POM-V.
Nobivac® KC contains live Bordetella bronchiseptica bacteria (strain B-C2) and canine parainfluenza virus (strain Cornell). POM-V.
Nobivac® L4 contains inactivated Leptospira strains: L. interrogans serogroup Canicola serovar Portland-vere (strain Ca-12-000), L. interrogans serogroup Icterohaemorrhagiae serovar Copenhageni (strain Ic-02-001), L. interrogans serogroup Australis serovar Bratislava (strain As-05-073) and L. kirschneri serogroup Grippotyphosa serovar Dadas (strain Gr-01-005). POM-V.
Nobivac® Lepto 2 contains inactivated Leptospira interrogans serogroups Canicola (strain Ca-12-000) and Icterohaemorrhagiae (strain 820K). POM-V.
Nobivac® Myxo-RHD PLUS contains live myxoma vectored RHD virus strain 009 and MK 1899. POM-V.
Nobivac® Parvo-C contains live attenuated canine parvovirus (strain 154). POM-V.
Nobivac® Pi contains live attenuated canine parainfluenza virus (strain Cornell). POM-V.
Nobivac® Rabies contains inactivated Rabies virus (strain Pasteur RIV). POM-V.
Nobivac® Respira Bb is an inactivated subunit vaccine containing the fimbrial antigen of Bordetella bronchiseptica. POM-V.
Nobivac® Tricat Trio contains live attenuated feline calicivirus (strain F9), feline herpes virus type 1 (strain G2620A) and feline panleucopenia virus (strain MW-1). POM-V.
Nobivac® Solvent contains sterile phosphate buffered water. POM-V
Further information is available from the SPC, datasheet or package leaflet.
Advice should be sought from the medicine prescriber.
Prescription decisions are for the person issuing the prescription alone.
Use Medicines Responsibly.
MSD Animal Health UK Limited, Walton Manor, Walton, Milton Keynes, MK7 7AJ, UK
Registered in England & Wales no. 946942





